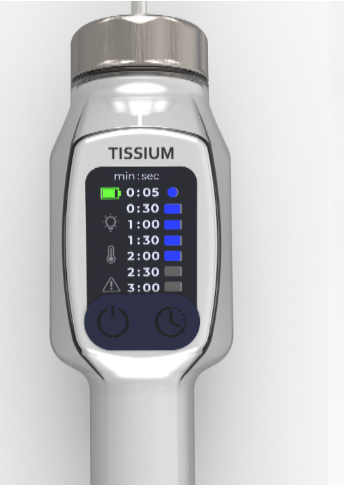
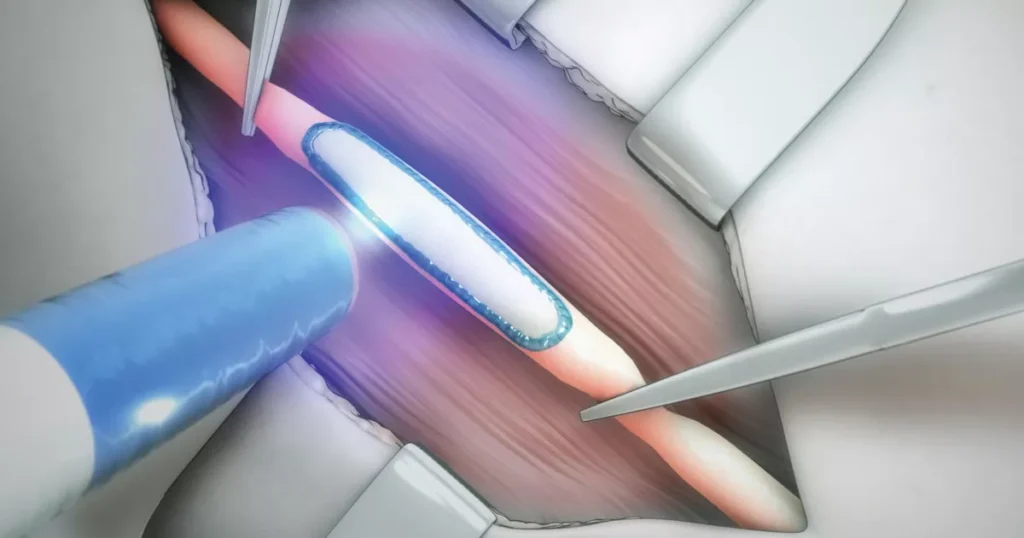
Support in sourcing and industrialization for a near UV light by Tissium
▪️The challenge
- Find reliable components, including electronic, mechanical and optic ones with medical quality standards and long time life, to ensure the assembled product will pass the certification ISO 13485 + CE and FDA and be able to be sold on the market
▪️ Solution
- Kickmaker has sourced different components supplier and realize a sourcing matrix including the cost, performance and quality, and realize test for the customers.
- Kickmaker recommended some components for the establishment of the final BOM of the customer.
▪️ Benefits
- Tissium could focus on their main activity of product and market development while Kickmaker ensured that the sourced components will be efficiently integrated in the manufacturing process, and will successfully pass the certification process to ensure Tissium to run its activity smoothly.
Technical development
For this significant Medtech project, Kickmaker supported Tissium in :
- Design and industrialization of the EVT prototype for the near-UV light source and charging station.
- Mechanical, electronic, and optical development to ensure the product’s performance and reliability in the medical field.
- Manufacturing validation to ensure that the final product meets medical standards and requirements.
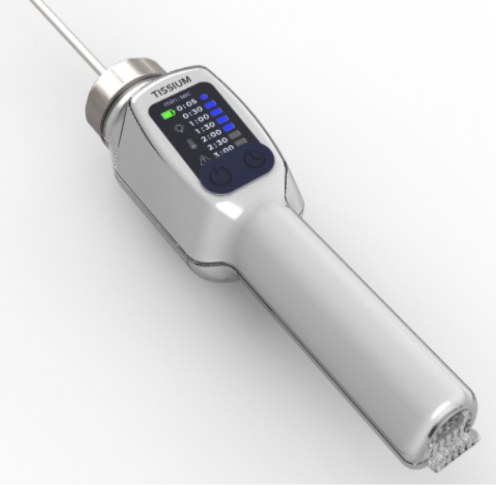
Kickmaker's expertise
Thanks to our expertise in the industrialization of medical products, we have enabled Tissium to:
- Integrated design of mechanical, electronic, and optical components to meet the needs of the medical industry.
- EVT prototyping and validation to ensure product compliance.
- Optimization of industrial production to ensure reliable, large-scale manufacturing.
Discover Kickmaker’s expertise
Innovation.
01. Innovation
design and industrialization.
Define the possibilities inherent in the development of your products and the associated industrial processes.
I innovate
Anticipation.
03. Management
product lifecycle management.
Production monitoring activities, quality control, and series life follow-up for your product.
I manage